Despite better technology, brands spend too much time on paper Certificates of Analysis (CoAs). As a result, CoAs are an essential building block for ensuring product quality and safety. They offer a glimpse behind the curtain at a supplier’s production process and detail the lot characteristics of each batch.
Paper CoAs are a problem
Despite their value, CoAs do little more than provide insight into one point in the supply chain. They’re merely a required document to be checked off by receiving. As such, a paper CoA does not tell the whole story of a supplier’s compliance with a manufacturer’s specifications. Nuance and potential ingredient variabilities are lost. Employees also tend to misplace, misfile, or ever lose paper CoAs, which can be catastrophic.
And even if everyone has a stake, paper CoAs prevent the efficient distribution of this mission-critical data.
Digital CoAs offer a solution
A simple digital solution for managing CoAs can transform a static quality control headache into a valuable tool that manufacturers can use to advance operational efficiencies and product outcomes.
Consider this:
- The supplier sends a digital CoA ahead of its assigned shipment.
- The manufacturer’s digital document management system retrieves it.
- The system stores the CoA in a centralized database, available instantly to every department.
- Internal stakeholders can connect production output to raw material attributes, and possible variances, in real-time.
- Automated workflows send alerts for items, raw materials, and ingredients that fall outside established parameters, eliminating waste while safeguarding quality.
A digital CoA approach also prevents disruptions, like production halts and recalls. When every department has instant access to the same information, manufacturers can proactively guard against risk — and lost time and resources.
Finally, with an integrated system in place, teams can track supplier performance in real-time and identify problems before they happen while ensuring quality and safety.
More than a static data point
Different departments leverage CoAs differently:
- Procurement can leverage CoAs to ensure suppliers are meeting specifications while also quantifying losses attributed to non-conformance. And the team can use this information in future supplier negotiations, when updating specs, or for other business-critical analyses.
- Quality can use CoAs to spot variabilities in shipment lots and compare individual data points to draw a more comprehensive conclusion about a supplier’s performance.
Streamlining and automating CoA administration lets a company quickly grade supplier performance, act quickly, cut unnecessary costs, eliminate lost production time, and improve supplier relationships.
Networked supplier compliance
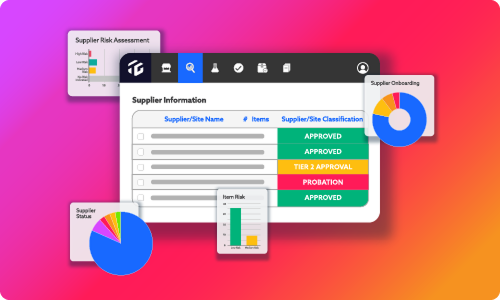
Networked Supplier Compliance evaluates supplier item, ingredient, and raw material performance, lot by lot, from purchase order to production. The software works as a virtual early warning system, identifying issues before supplier shipments leave the shipping dock.
Teams can scorecard, measure, and rank suppliers by risk category, providing an intuitive, visual overview of supplier performance. In addition, downstream issues like customer complaints and plant floor problems can be traced to specific supplier lot shipments, allowing supplier chargebacks and rapid replacement. Most importantly, Networked Supplier Compliance stores the data in a centralized location, allowing teams to review specs, track CoA trends, and run reports on any critical data with one click.
Check out this on-demand webinar for a deep dive into our industry-standard forms and extensive attribute library, and discover how supplier data and business rules inform specification data, automate supplier compliance, and eliminate supplier and ingredient risk.