In this penultimate chapter of our limited Coffee Talk series, Regulatory Standards Manager Kari Barnes highlights the importance of standard forms.
TraceGains: What are TraceGains standard online forms?

Kari Barnes: They’re a godsend. Let me explain by first providing a little background.
I was a TraceGains end-user before I became an employee. I worked on a small supplier approval team for a food ingredient manufacturer with 100 global manufacturing and distribution locations. We were managing about 90 suppliers, 330 items, supplier approvals, supplier monitoring. And the never-ending, brutal matrix of maintaining supplier documentation by manually slogging through each process individually. It was an overwhelming, arduous, and error-prone process that often led to expired and missing documents. Suffice to say, there were multiple opportunities for improvement. There was a world of difference managing these processes before and after TraceGains. I’m absolutely not kidding when I say that I’m one of our company’s biggest advocates.
TG: Now you’re on the other side, managing these online forms. Tell us about them.
KB: TraceGains worked with about 50 companies of all sizes and industry verticals, and landed on the most common documents in the food, beverage, and dietary supplement industries. With input from all these different companies, TraceGains was able to standardize and normalize the data elements required on each document-type for compliance with GFSI and FSMA requirements, nutrition and supplements product labeling, as well as the SIDI [Standardized Information on Dietary Ingredients] initiative.
From this, TraceGains then developed standard online forms. These included supplier and item questionnaires, nutrition, allergens, suitability, sustainability, foreign supplier verification program, and national bioengineered food disclosure forms. Then we convinced our customers to start using these in place of each company’s proprietary document. Since we established this standardization, market-wide adoption continues to this day.
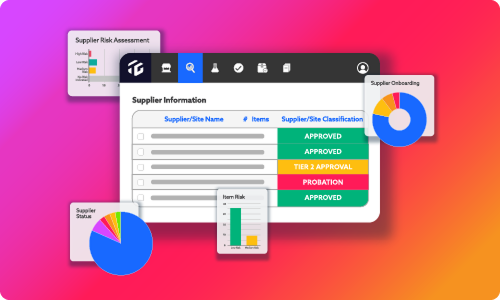
But the value of these forms is not just this standardization across industry, but the actionable data PDFs or printouts can’t provide. Since everything is digital, users can leverage the data for dashboarding, reporting, and automated risk scoring. We upload the data automatically with each new document submission so users have an up-to-date, clear snapshot of suppliers and items.
TG: But what about those who need to complete these standard forms?
KB: The standardized online forms benefit both suppliers and manufacturers. For suppliers, instead of filling out a form for every customer, they can fill it out once and post it to all their customers on TraceGains Network. And if a company doesn’t have all its customers using TraceGains, they can just download the completed form as a PDF and send that to any other customer anywhere in the world. Then, the next time someone needs to complete that form, rather than filling out a new one, the supplier has to update a few fields and resubmit it. And that saves everyone time.
For manufacturers, the forms create streamlined compliance with GFSI and FSMA requirements via automated supplier documentation and approvals. The forms automate much of the work around these processes and seamlessly guides users through the rest. In short, the forms allow suppliers and manufacturers to spend less time and resources managing these processes. Everyone can get back to generating revenue for their business rather than being paper clerks, which is a degree no one went to school for.
TG: Speak a little more about the value of data vs. just documents.
KB: OK, but let me address automation first. As I said before, supplier documentation is a never-ending, brutal matrix to maintain. That’s not hyperbole. The number of documents required from a single supplier can be staggering. Step one is getting all the documents, if you’re lucky. Step two is maintaining them and keeping them up-to-date, which is infinitely more complicated because every document expires at a different interval and frequency. It’s messy. And painful. And so time consuming.
With the standard online forms, users can send out document requests en masse or individually over TraceGains Network, and stored in one place. The system automatically notifies suppliers about missing or expired documents until they fulfill them. The processes of having and using the standardized online forms streamlines the complexities of managing and maintaining up-to-date supplier documentation.
The standard online forms also automate the supplier approval process to help you make faster and better-informed decisions. The answers provided on the standard online forms can be pulled into dashboards and reports. The dashboards and reports are automated, scalable, and configurable and updated with every new document submission. Your team gets an immediate, up-to-date view of supplier performance at the corporate, production, raw material, and packaging levels.
You can then begin the work you’ve never really had time for managing by exception, because the system will alert you. For example, with the time you save on the supplier approval process, you can fortify your supply chain by qualifying backup suppliers. And because the supplier data is updated with each new document submission, you can see and react to changes in-real time.
So, what are SOFs and what are their benefits? They’re amazingly insightful tools that create order out of chaos. They create better and more meaningful compliance to GFSI and FSMA. They create streamlined and efficient processes for maintaining supplier documents, supplier approval, and monitoring supplier performance.
And I’m working on making all of this available globally, with regulatory compliance standards and standard forms for other jurisdictions.